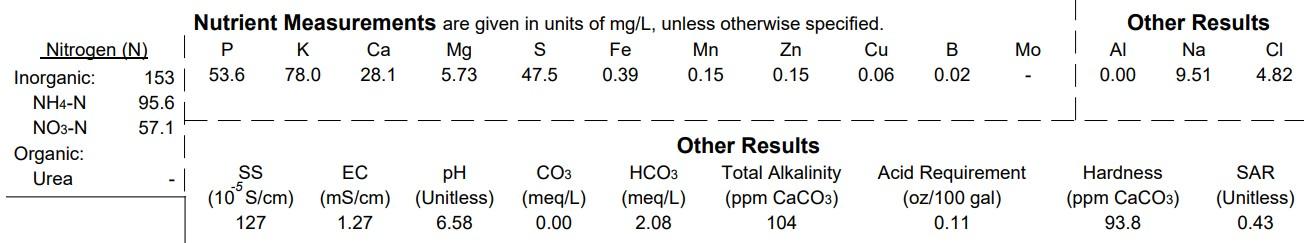
NItrogen (N)
Total concentration of inorganic nitrogen and the individual concentrations of inorganic nitrate-nitrogen (NO3-N) and ammonia-nitrogen (NH4-N). Results are reported in mg/L which is equivalent to parts per million (ppm).
Other Nutrients
Concentrations of phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulfur (S), iron (Fe), manganese (Mn), zinc (Zn), copper (Cu), and boron (B) are reported in mg/L which is equivalent to parts per million (ppm).
Other Results
Sodium (Na) and chloride (Cl) are elements that may be important for plant nutrition but are more often detrimental to plant health in excessive levels.
Aluminum (Al) is not a plant nutrient and may create toxicity when soil or substrate pH is excessively low. Aluminum also plays a role in coloration for ornamental crops such as hydrangea.
EC (Electrical Conductivity) is reported in units of mS/cm. It measures the total quantity of dissolved salts (ions) in solution. It may be used as a general evaluation of the total nutrient concentrations available to plants. However, it also includes elements such as sodium (Na) and chloride (Cl), which increase EC without providing nutritional value. In addition, excessively high EC will burn plant roots regardless of the whether the ions are required for plant nutrition.
SS (Soluble Salts) is the same as EC, but is expressed in units of 10-5 S/cm.
The pH is the level of acidity or basicity as measured by the amount of hydrogen ions (H+ ) in solution. It ranges from 0 (acidic) to 14 (basic) on a logarithmic scale; 7 indicates neutrality. It can reduce the availability of micronutrients (Fe, Mn, Zn, Cu, B) when pH is high (>6.5) and the availability of macronutrients (N, P, K, Ca, Mg, S) when pH is low (<5.0). It also impacts the efficacy of pesticides.
Total alkalinity (CO3 + HCO3 ) is a measure of water's ability to neutralize acids or resist change in pH (pH buffering capacity). It is reported as ppm calcium carbonate (CaCO3 ). It indicates the tendency of irrigation water or nutrient solutions to increase soil or substrate pH. In some plant production systems, alkalinity must be neutralized to avoid increasing substrate pH to levels that reduce micronutrient availability or do not support plant growth.
Acid Requirement (AR) indicates the number of ounces of battery acid (35% sulfuric acid) to apply to each 100 gallons of water to lower alkalinity to a desirable level. The AR on the report is the amount required to neutralize 100% of the alkalinity above 100 ppm CaCO3 in the water sample. If you would like to neutralize with a different acid, this acid calculator can be used.
Hardness indicates the amount of Ca in water, expressed as ppm of calcium carbonate (CaCO3). It is useful in evaluating water for potential to build up scale or for compatibility with animal medicines.
SAR (Sodium Adsorption Ratio) indicates the ratio of Ca + Mg: Na. It is useful in predicting the potential of Na to accumulate in the soil or soilless media and damage plant root systems. Ca and Mg may ameliorate the negative effects of Na in soil or substrate systems. Generally, SAR values >10 indicate the potential for Na damage to plant health.
This page was last modified on 05/12/2026
