- Submit plant samples in PAPER bags or envelopes. Undried samples submitted in plastic bags are prone to decay in transit.
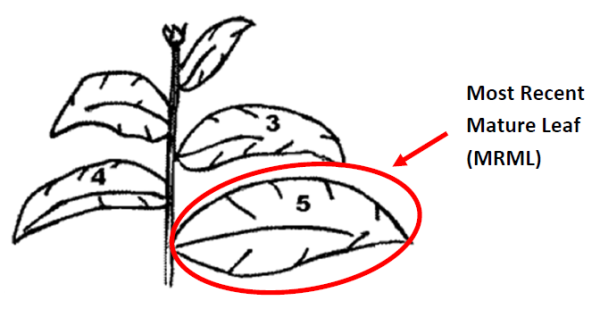
- Unless otherwise specified in the NCDA&CS Plant Tissue Analysis Guide, the correct plant part for most crops is the most recent mature leaf (MRML).
- To obtain a complete nutrient analysis, submit enough plant material. A general rule of thumb is to collect at least two handfuls of leaves.
For detailed information, please refer to the NCDA&CS Plant Tissue Analysis Guide.
Plant Part Codes and Descriptions
| Code | Description | Example Crop |
|---|---|---|
| M | Most recent mature leaf (MRML), remove petiole during sample collection | Most plants, including cotton & strawberry (include separated petioles with leaf sample for these 2 crops) |
| W | Whole plant (cut 1/2 to 1" above soil surface, including roots will affect results) | Seedlings or young plants |
| T | Top 3 - 6 inches or top 2 - 4 leaves | Turf; forage grass and small grains prior to reproductive growth stages |
| E | Ear leaf (opposite and below ear) | Corn (from tasseling through silking) |
| P | Petiole only | Vinifera Grape |
| H | Harvest leaf | Tobacco |
Sampling Quick Guides for Specific Crops
When: Diagnostic (troubleshooting) samples can be collected any time during the growing season as long as some primocanes are present. Collect a “good” (healthy) and a “bad” (unhealthy) sample.
Predictive samples evaluate nutritional needs for next year’s crop and should be collected from primocanes post-harvest and before topping.
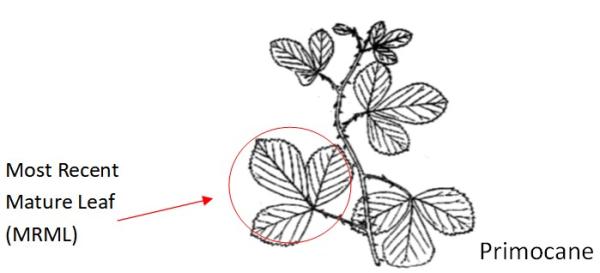
How: Collect leaves from primocanes; 1–2 leaves per bush from 20–40 bushes; 20-40 leaves total
Plant part: Collect Most Recent Mature Leaves (MRML) from primocanes. MRMLs are neither young and shiny nor old and dull. About the 5th leaf down from the tip.
When: For predictive (routine) testing, sample plants after fruiting. Diagnostic (troubleshooting) samples may be collected at any growth stage.
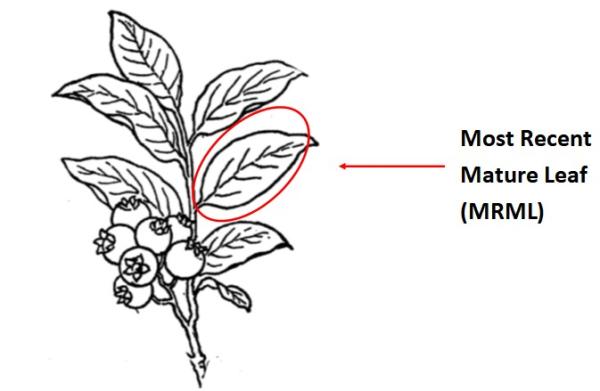
How: Collect 1–2 leaves per bush; 30-50 leaves total or about two handfuls. Collect mature leaves from mid-portion of current season's growth.
Plant part: Collect Most Recent Mature Leaves (MRML) from mid-shoot area. MRMLs are neither young and shiny nor old and dull. About the 4-5th leaf down from the terminal bud.
When: Predictive and diagnostic samples may be collected at anytime during the growing season prior to silking.
Important: Tissue sampling after silking is not recommended. Fertilizer applications after this stage have not shown consistent yield or economic increases.
| Growth Stage | Growth Stage Code | Plant Part | Plant Part Code | Leaves to Collect |
|---|---|---|---|---|
Seedling (<4") Early (4-12") | Seedling (S) Early (E) | Entire top of plant cut 1" above soil | Whole (W) | 15-20 plants |
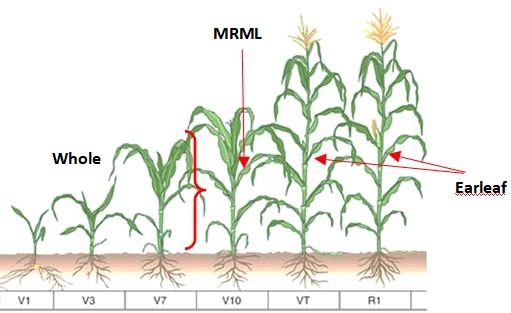
| Prior to tasseling (>12") | Early (E) | First fully developed leaf below the whorl. Leaf should be totally unrolled and developed sheath (collar) on the stalk. | Most Recent Mature Leaf (M) | 10-15 |
| Tasseling (VT) to silking (R1) | Bloom (B) Fruit (F) | Leaf opposite and below the uppermost developing ear | Earleaf (E) | 10-15 |
Important: All cotton samples receive petiole nitrate analysis. Detach petioles from leaves, band together, and place in same paper bag as corresponding leaf samples.
Collect 25-30 most recent mature leaves (M) and petioles.
| Growth Stage Code | Growth Stage | Weeks |
|---|---|---|
| Seedling (S) | Four weeks following emergence of 2-3 true leaves | 1-4 |
| Early (E) | Four weeks following seedling stage, includes pinhead square formation | 1-4 |
| Bloom (B) | Plants have at least 5 open blooms per 25 row feet | 1-4 |
| Fruit (F) | 5th week after beginning of bloom | 1-4 |
Important: Specify whether this is a Field grown cucumber (in soil) or a greenhouse cucumber (soilless media) as this will determine your recommendations.
Note for vining crops: The most recent mature leaf may be further down than vine than the 4th or 5th leaf
| Crop | Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|---|
| Field Cucumber (Soil) | Early to Fruiting | Early (E), Bloom (B), Fruit (F) | Most recent mature leaf (M) | 15-20 |
| Greenhouse Cucumber (Soilless) | Early to Fruiting | Early (E), Bloom (B), Fruit (F) | Most recent mature leaf (M) | 8-10 |
Important: The Plant Tissue Lab analyzes forages for monitoring crop fertility. Forage hay samples analyzed for animal nutrition should be sent to the Food & Drug Protection Division: Animal Feed Program. Located at: 4400 Reedy Creek Road, Raleigh, North Carolina 27607.
| Growth Stage | Growth Stage Code | Plant Part | Plant Part Code | Leaves to Collect |
|---|---|---|---|---|
| Tillering (<6" tall) | Seedling (S) | Entire plant cut 1/2 inch above soil | Whole (W) | 2 handfuls |
| >6" Tall, prior to seed head (boot) formation | Early (E) | Top 6 inches of plant or upper half (top 4 leaves) | Top (T) | 20 tops |
| After seed head formation (Diagnostic only) | Fruit (F) | Leaf below seed head | Most recent mature leaf (M) | 20-30 leaves |
Important: Specify the grass species on the submission form.
When: For predictive (routine) testing, sample plants during dormancy—after bud set (August-September) and before bud break (February-March). September is ideal. Diagnostic (troubleshooting) samples may be collected at any growth stage.
Growth Stage: Early (E) or Mature (M)
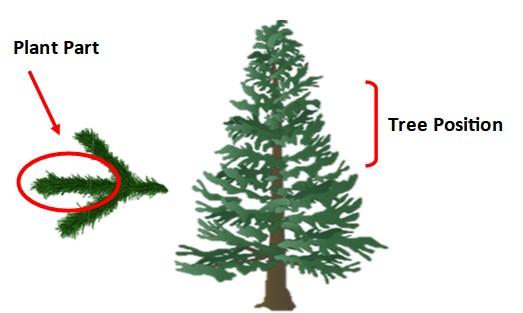
Tree Position: Collect shoots from the current season's growth from the upper half of the tree. Do not sample from the top whorl.
Plant part: Most Recent Mature Leaf (MRML). Break off 4-6”mature lateral shoots from 10–20 trees for ~10-20 shoots total.
When: Mid to late summer but prior to final fruit swelling. Diagnostic (troubleshooting) samples may be collected at any growth stage.
How: Sample each variety separately. Collect 25 to 60 most recently mature leaves (MRMLs) located opposite the flower/fruit clusters on well-exposed shoots. Generally, the 1st or 2nd fruit cluster from the base of the shoot. Do not collect more than one or two MRMLs per vine.
Important: Detach leaves from petioles in the field and submit leaves only.

When: For predictive (routine) testing, sample plants after full bloom through veraison (fruit ripening). Diagnostic (troubleshooting) samples may be collected at any growth stage.
How: Sample each variety separately. Collect 1–2 petioles per vine; 25-30 petioles total or about two handfuls.
Plant part: Petioles should come from most recent mature leaves opposite fruit clusters on well-exposed shoots, generally the 1st or 2nd fruit cluster from the base of the shoot.
Important: Detach petioles from leaves in the field and submit petioles only.

When: Recommended sampling time for nutritional testing is from early vegetative growth up until flowering.
How: Collect 1-2 leaves from 10-20 plants.
Plant Part: Sample the most recently mature leaves (MRMLs), generally the 3rd to 5th leaf from the growing point.
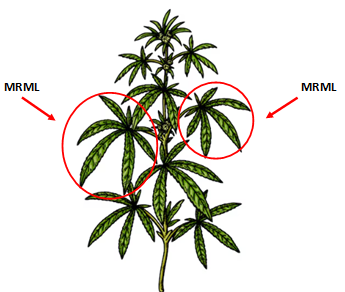
When: For predictive (routine) testing, sample plants after fruiting. Diagnostic (troubleshooting) samples may be collected at any growth stage.
How: Collect leaves near base of current year’s growth; 1–2 leaves per tree from 20–30 trees; 20-40 leaves total.
Plant part: Collect most recent mature leaves (MRML) from mid-shoot area. MRMLs are neither young and shiny nor old and dull. About the 5th leaf down from the terminal bud.
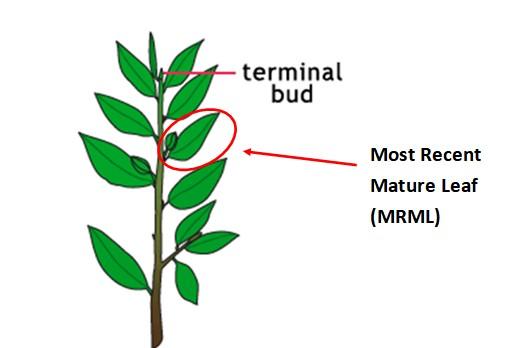
When: Early growth through bloom.
Plant part: Most recently mature tetrafoliate leaf (MRML) which is about 3rd to 5th leaf from growing point.
How: 25-30 leaves from 15-20 plants. Detach petioles in the field and submit leaves only.

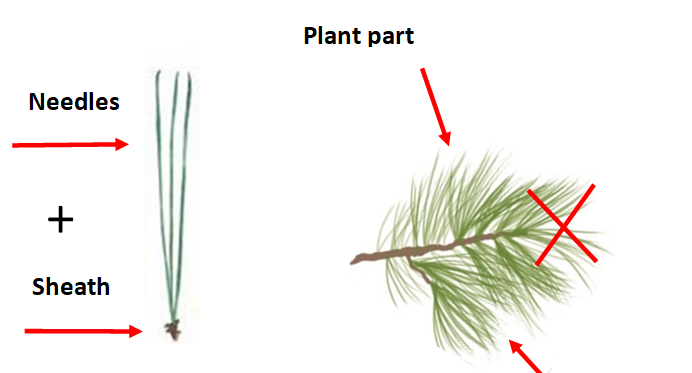
When: Collect routine (predictive) samples from May to June. Diagnostic (troubleshooting) samples may be collected at any time.
Tree Position: Middle of the tree.
Quantity: Collect 2 handfuls of needles (~50).
Plant part: Collect needles with sheaths from the lateral areas of the bough. This is the Most Recent Mature Leaf (MRML). Do not collect needles from the tufted ends.
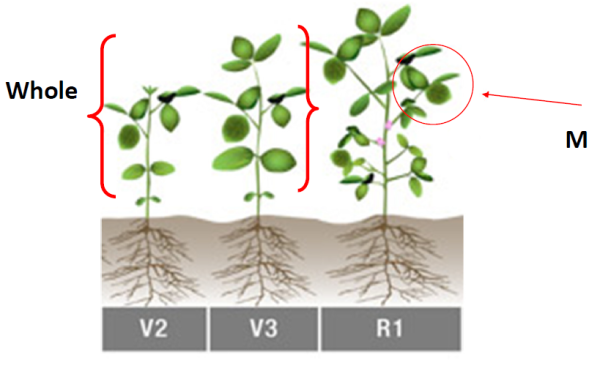
Tissue analysis is used to optimize soybean fertility (predictive) or troubleshoot nutritional problems (diagnostic) from seedlings (V2) up to R2. Sampling after R4 is not recommended.
| Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|
| Seedlings (less than 12" tall), V2-V3 | Seedling (S) | Whole (W) | 20-30 |
| Prior to bloom, >V3 | Early (E) | Most Recent Mature Leaf (M) | 20-30 |
| Initial bloom to before pod set, R1-R2 | Bloom (B) or Fruit (F) | Most Recent Mature Leaf (M) | 20-30 |
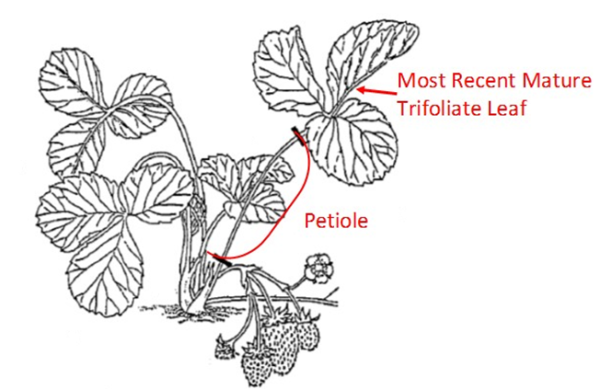
Important: All strawberry samples receive petiole nitrate analysis. Detach petioles from the trifoliate leaf, band petioles together, and place in same paper bag as corresponding leaves.
In-season monitoring: Begin collecting tissue samples when spring growth begins and continue every 1-2 weeks throughout blooming and fruiting stages. To monitor the nutrient needs beginning the first week of bloom. This is generally 1 week after the 1st spring nitrogen application and then every 2 weeks for 12 weeks.
Week: This is the number of weeks since first bloom. The first week of Bloom is week 1. The first week of fruit is about week 6. The week count does not reset when the plant moves into fruiting stage.
| Production System | Growth Stage Code | Approximate Weeks | Plant Part Code | Leaves to Collect |
|---|---|---|---|---|
| Field | Early (E) | Week 1-8 | Most Recent Mature Leaf (M) | 20-25 |
| Bloom (B) | Week 1-6 | Most Recent Mature Leaf (M) | 20-25 | |
| Fruit (F) | Week 6-12 | Most Recent Mature Leaf (M) | 20-25 | |
| High Tunnel | All Growth Stages (E, B, F) | Use Field Weeks | Most Recent Mature Leaf (M) | 20-25 |
| Greenhouse (Soilless) | All Growth Stages (E, B, F) | Use Field Weeks | Most Recent Mature Leaf (M) | 20-25 |
Predictive and Diagnostic Testing
When: Throughout the growing season. For routine monitoring, 1 week before layby is best.
| Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|
| Seedlings, Greenhouse transplants | Seedling (S) | Whole (W) | 1 Tray |
| Before bloom | Early (E) | Most Recent Mature Leaf (M) | 8-12 |
| Early bloom | Bloom (B) | Most Recent Mature Leaf (M) | 8-12 |
| Late bloom | Fruit (F) | Most Recent Mature Leaf (M) | 8-12 |
| Mature | Mature (M) | Most Recent Mature Leaf (M) | 8-12 |
Harvest Ripeness Testing
When: At plant maturity, before harvest.
| Position Description | Position Code | Plant Part Code | Leaves to Collect |
|---|---|---|---|
| Upper leaves (tips), 21st to 30th nodes from the bottom | Upper (U) | Harvest Leaf (H) | 8-12 |
| Middle leaves (smoking), 11th to 20th nodes from the bottom | Middle (M) | Harvest Leaf (H) | 8-12 |
| Lower leaves (lugs & cutters), 1st to 10th node from the bottom | Lower (L) | Harvest Leaf (H) | 8-12 |
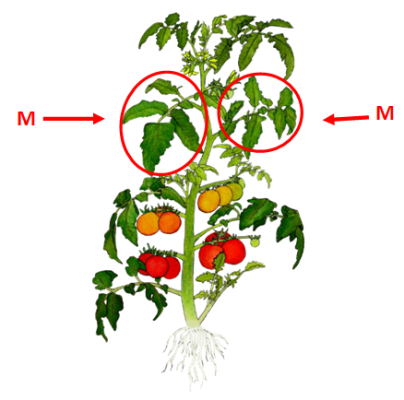
How: Collect 1-2 leaves per plant to make a representative sample of about 10 to 15 leaves total. Collect the most recent mature leaf (M).
Important: Specify whether this is a Field grown tomato (in soil) or a greenhouse tomato (soilless media) as this will determine your recommendations.
Field Tomato (Soil)
| Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|
| Early growth (5-leaf stage through first flower) | Seeding (S) or Early (E) | Most Recent Mature Leaf (M) | 8-10 |
| Early flower through first fruit set (golf-ball sized fruit) | Bloom (B) | Most Recent Mature Leaf (M) | 8-10 |
| First fruit set through harvest | Fruit (F) or Mature (M) | Most Recent Mature Leaf (M) | 8-10 |
Greenhouse Tomato (Soilless Media)
| Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|
| Early growth through first fruit set | Seedling (S), Early (E), or Bloom (B) | Most Recent Mature Leaf (M) | 8-10 |
| First fruit set through harvest | Fruit (F) or Mature (M) | Most Recent Mature Leaf (M) | 8-10 |
Predictive and Diagnostic Sampling
When: Sample during the growing season either for routine monitoring (predictive) or troubleshooting problems with turf health (Diagnostic).
How: Collect all turf clippings from area of interest (can be collected directly from mower basket). Mix together and remove 1 pint of tissue as a subsample. Allow to air dry and remove any non-turf debris.
| Growth Stage | Plant Part | Leaves to Collect |
|---|---|---|
| Early (E) | Top (T) | 2 handfuls |
| Mature (M) | Top (T) | 2 handfuls |
Diagnostic (Troubleshooting) sampling can be done at any growth stage and does not require a biomass sample.
Predictive (GS-30) sampling is used to determine mid-season N rates and evaluate overall nutritional status.

When: Zadoks growth stage 30 (GS-30) or Feekes 5. This is about February to March but the correct growth stage is critical.
When wheat begins to stand up tall and straight, pull several plants, split the stems, and look at the growing point. Before GS-30, the growing point will be just above the roots. At GS-30, the growing point is about 1/2 inch up the stem.
How: GS-30 sampling requires two types of samples. Submit in separate paper bags labeled with sample ID and tissue or biomass, respectively.
| Sample Type | Instructions |
|---|---|
| Tissue | Cut plants about 1/2 inch above surface. Two large fistfuls make a good sized sample. Growth stage = S. Plant Part = W |
| Biomass | Collect all above-ground plant tissue in a 36" row section or from 1 square yard (broadcast fields). |
| Growth Stage | Growth Stage Code | Plant Part | Leaves to Collect |
|---|---|---|---|
| Seedling to early jointing, GS 3-6 (Feekes), GS 26-21 (Zadoks) | Seedling (S) | Whole (W) | 2 handfuls |
| Early jointing to just prior to heading, GS 7-9 (Feekes), GS 32-39 (Zadoks) | Early (E) | Top (T) | 25-40 |
| Just prior to heading, GS 10-11 (Feekes), GS 45-100 (Zadoks) | Bloom (B) | Flag Leaf (M) | 30-40 |

This page was last modified on 05/12/2026
